When Does Food Become a Drug? It sounds like a simple question. But it isn’t.
At what point does something traditionally consumed as food become regulated as a drug?
The answer has less to do with chemistry and more to do with classification.
The Role of Intention
In regulatory frameworks, the distinction between food and drugs often hinges on intended use.
- A food is consumed for nourishment.
- A drug is intended to diagnose, treat, cure, or prevent disease.
The compound itself may not change.
But the claims surrounding it do.
When a substance begins to be marketed with disease-specific language, regulatory status shifts.
That shift can reshape public perception — sometimes dramatically.
When Does Food Become a Drug?
Same Molecule, Different Category
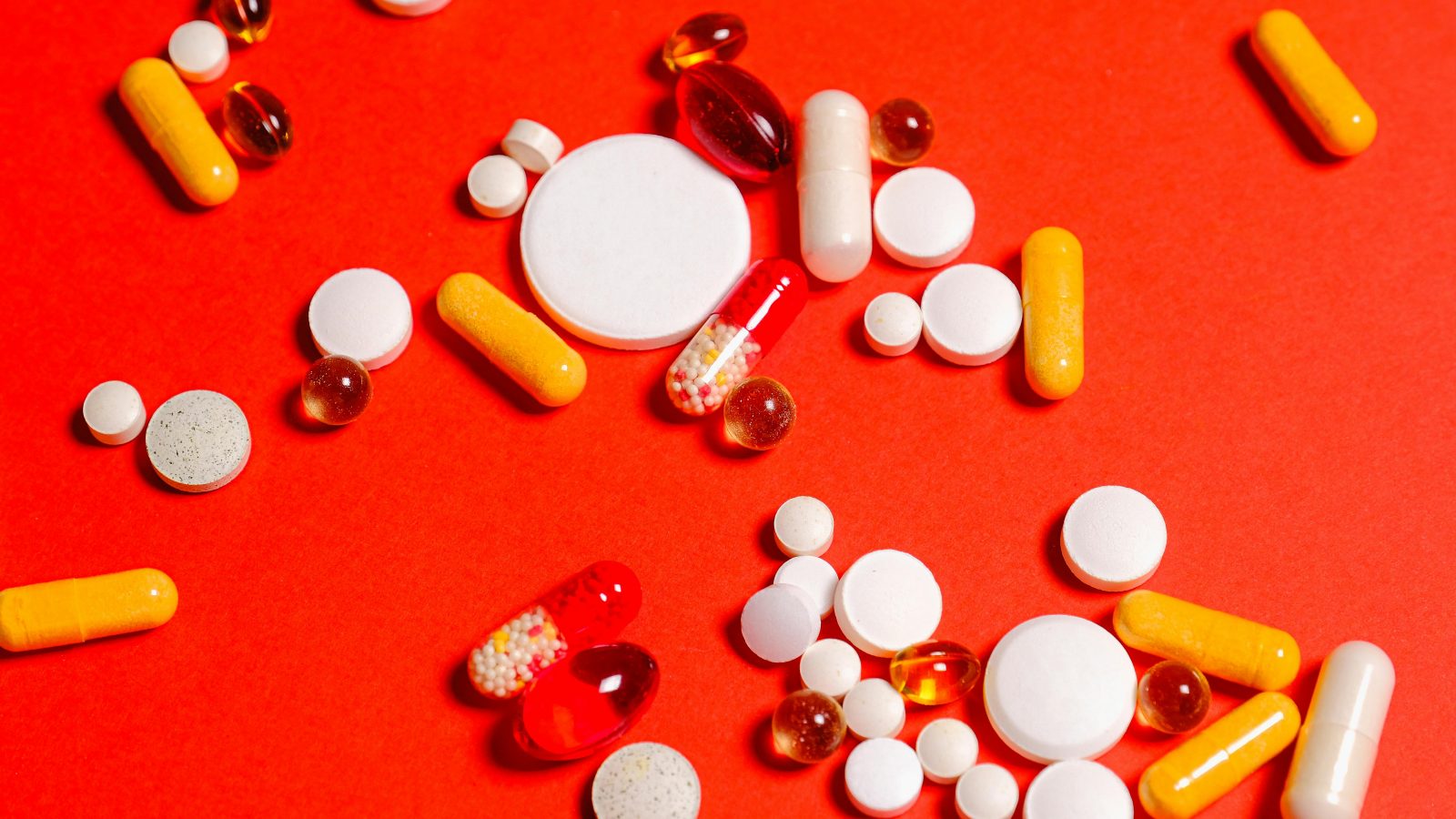
Many compounds exist in both food and pharmaceutical contexts.
- Caffeine appears naturally in coffee beans and tea leaves. It also appears in over-the-counter medications.
- Melatonin is produced in the body and found in foods. It is also sold as a supplement.
- Omega-3 fatty acids occur in fish. They are also packaged into concentrated capsules.
The molecule doesn’t fundamentally change.
Its regulatory category can.
The Power of Framing
When a substance is framed as part of daily nourishment, it occupies one mental category.
When it is framed as a treatment, it moves into another.
This framing affects:
- How the public perceives risk
- How regulators classify it
- How the media discusses it
- How consumers approach it
Once something crosses into the “drug” category, it carries assumptions of danger, potency, and medical oversight, even if its chemical structure remains identical to what was previously consumed as food.
The Historical Tension
Throughout the 20th century, several naturally occurring compounds entered regulatory gray zones when their intended use shifted.
In some cases, food-derived substances were reclassified once therapeutic claims became central to marketing language.
The result was often confusion. Was the compound itself inherently different? Or had its framing changed? These are not simple legal debates. They shape how entire generations think about certain nutrients.
Why This Matters Today
As interest in traditional foods and bioactive plant compounds resurfaces, understanding regulatory classification becomes essential.
- If a compound is discussed within the context of nourishment and dietary diversity, it occupies one space.
- If it is promoted as a disease intervention, it occupies another.
The distinction is not trivial. It influences research funding, media coverage, enforcement priorities, and consumer trust.
Literacy Over Reaction
Rather than reacting emotionally to labels, a more productive approach is to understand the framework.
- What is being claimed?
- How is it being positioned?
- Is the conversation about nutrition — or about treatment?
Clarity on those questions prevents confusion.
And it allows adults to engage thoughtfully with both food and policy.
Returning to First Principles
- Food has historically been the foundation of health.
- Drugs have historically been interventions.
When the lines blur, tension arises, not because molecules change, but because language does.
Understanding that distinction does not require controversy. It requires literacy. When food remains food, the conversation stays grounded.
Want to Learn More?
📘 Download the Book, World Without Cancer: The Story of Vitamin B17 by G. Edward Griffin — Free PDF available.
🌱 Explore Natural Options and Receive a 10% Discount: Learn about Laetrile, B17, and Apricot Seeds at https://RNCstore.com/WLT.
🌍 Join the Movement: Visit Operation World Without Cancer to support research, education, and advocacy for natural healing.
💧 Find a Wellness Provider: Visit B17works.com to connect with a Richardson Certified Provider.

Join the conversation!
Please share your thoughts about this article below. We value your opinions, and would love to see you add to the discussion!